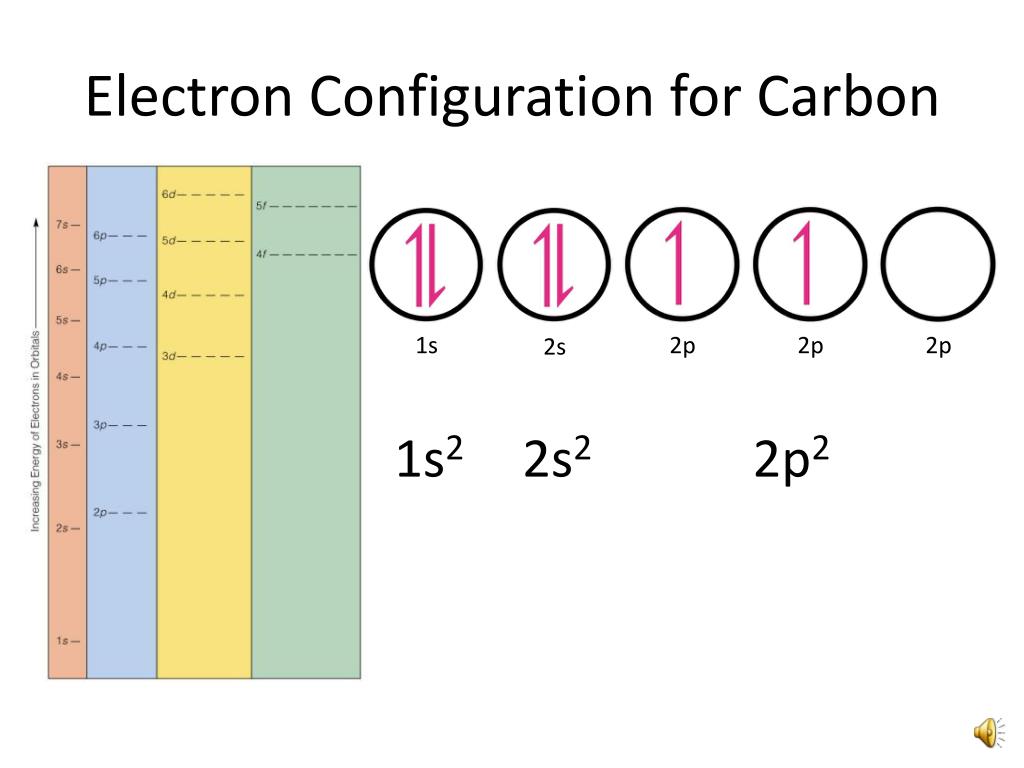
Electron Configuration Of Carbon
An electrochemical conversion of carbon dioxide into chemical fuels is a promising approach to store the renewable energy sources 2. However, a critical challenge toward efficient CO 2 reduction.
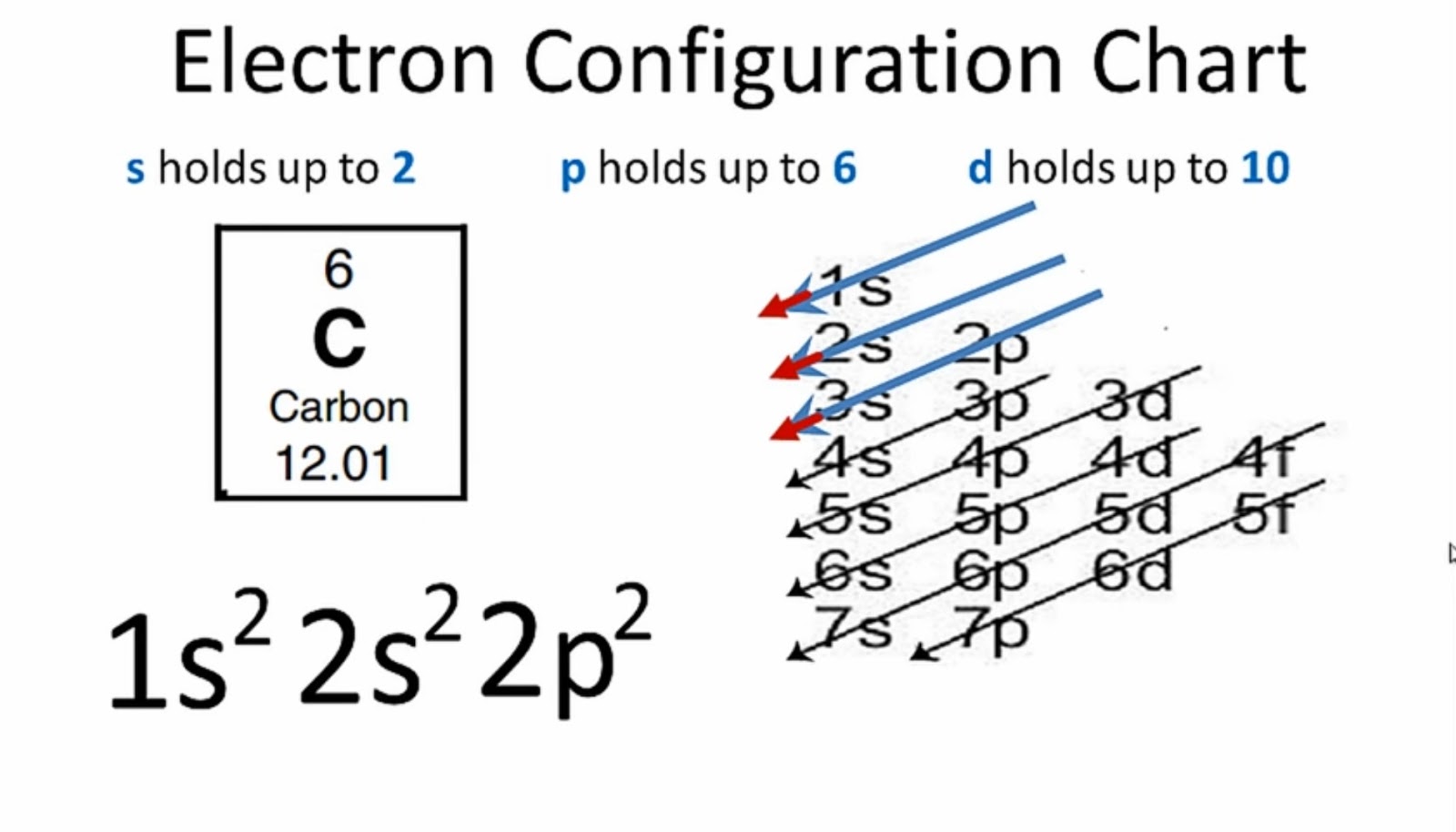
What Is the Carbon(C) Electron Configuration?
The electron configuration of an atom of any element is the of electrons per sublevel of the energy levels of an atom in its ground state . This handy chart compiles the electron configurations of the elements up through number 104.

Orbital Diagram For Cobalt
1. Introduction Electron configuration describes the distribution of electrons within an atom. The concept of electron configuration has been introduced since the discovery of Bohr atomic model. However, the electron configuration referred in this study was one that derived from the later atomic model - the quantum mechanics atomic model.
Electron Configurations Chemistry Steps
What is the electron configuration of cobalt? The total number of electrons in cobalt is twenty-seven. These electrons are arranged according to specific rules in different orbitals. The arrangement of electrons in cobalt in specific rules in different orbits and orbitals is called the electron configuration of cobalt.

Electron Geometry for CO2 (Carbon Dioxide) YouTube
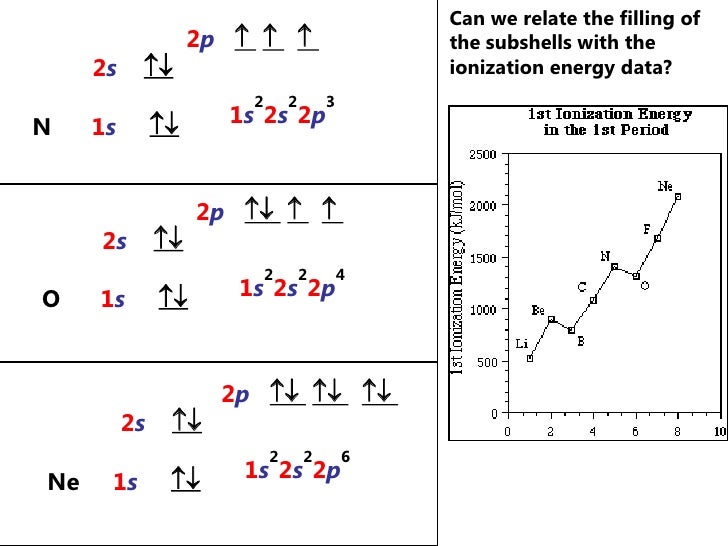
Let us return to the electron configuration of nitrogen and write it again: Find the atomic number of nitrogen (7) and use this electron configuration calculator to get a complete electron configuration. Otherwise, write the order of the energy levels with electron configuration chart: 1 s 2 2 s 2 2 p 3 \rm 1s^22s^22p^3 1 s 2 2 s 2 2 p 3.

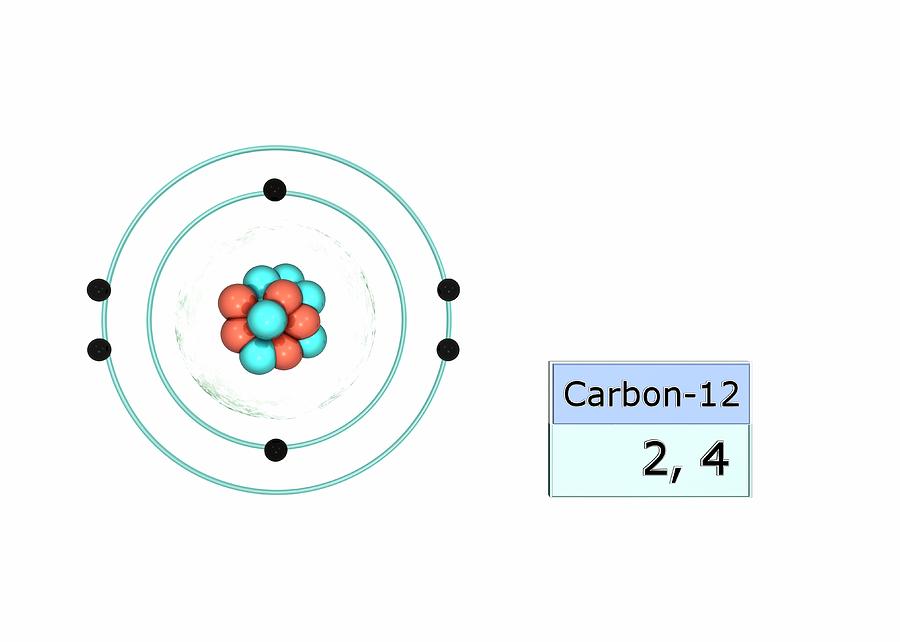
Carbon Electron Configuration
Here, the electron configuration of cobalt ion(Co 2+) is 1s 2 2s 2 2p 6 3s 2 3p 6 3d 7. This cobalt ion(Co 2+) has twenty-seven protons, thirty-two neutrons, and twenty-five electrons. Also, cobalt has one more ion. That is Co 3+. Co - 3e - → Co 3+ Here, the electron configuration of cobalt ion(Co 3+) is 1s 2 2s 2 2p 6 3s 2 3p 6 3d 6.

So far, we’ve used 16 of the CO2 Lewis structure’s total 16 outermost
The electron configuration of Co is 1s2 2s2 2p6 3s2 3p6 3d7. Co requires three more valence electrons in its d-orbital to attain full filled configuration. What is the complete electron configuration of Co 2+? The complete electron configuration of Co2+ is 1s2 2s 2 2p6 3s2 3p6 3d7.Co2+ have 7 valence electrons in the subshell.
13+ Co2 Dot And Cross Diagram Robhosking Diagram
PROBLEM 3.1.13 3.1. 13. Thallium was used as a poison in the Agatha Christie mystery story "The Pale Horse.". Thallium has two possible cationic forms, +1 and +3. The +1 compounds are the more stable. Write the electron structure of the +1 cation of thallium. Answer.
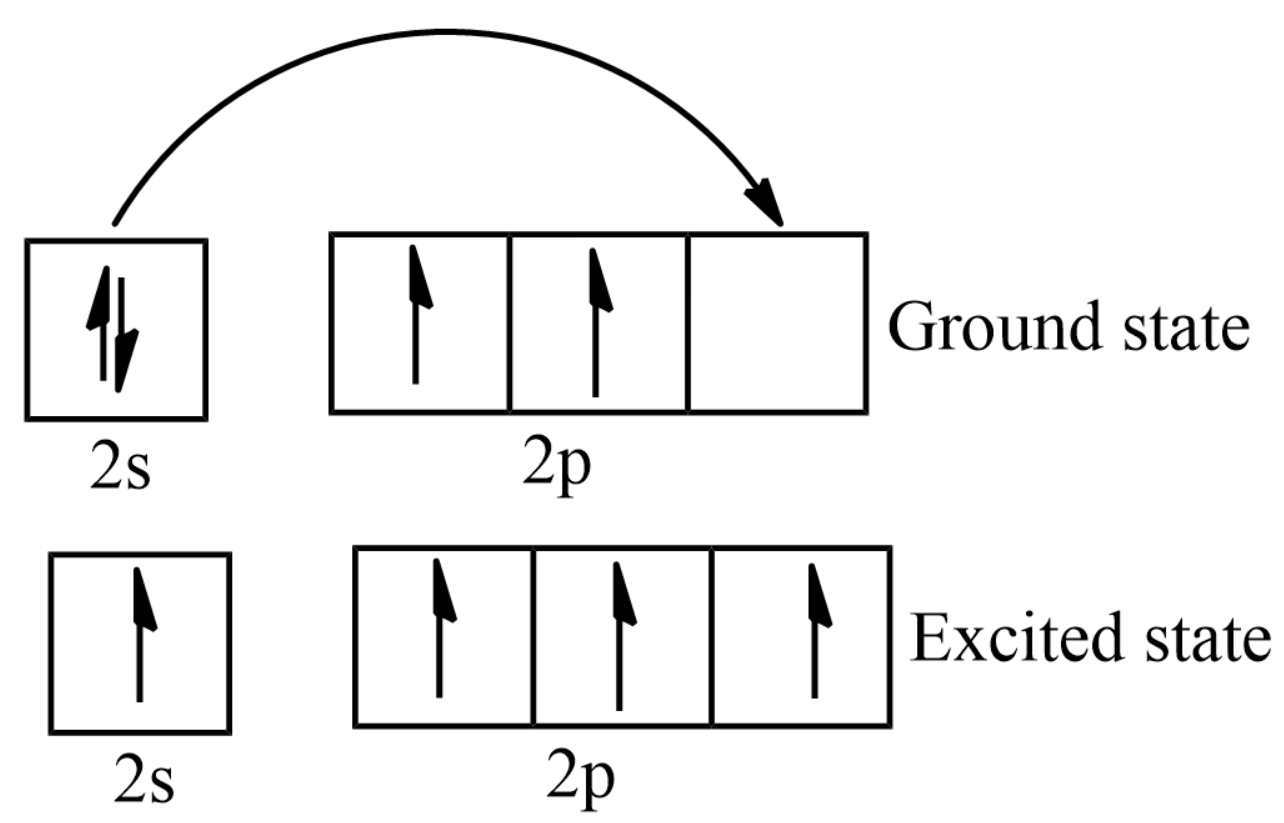
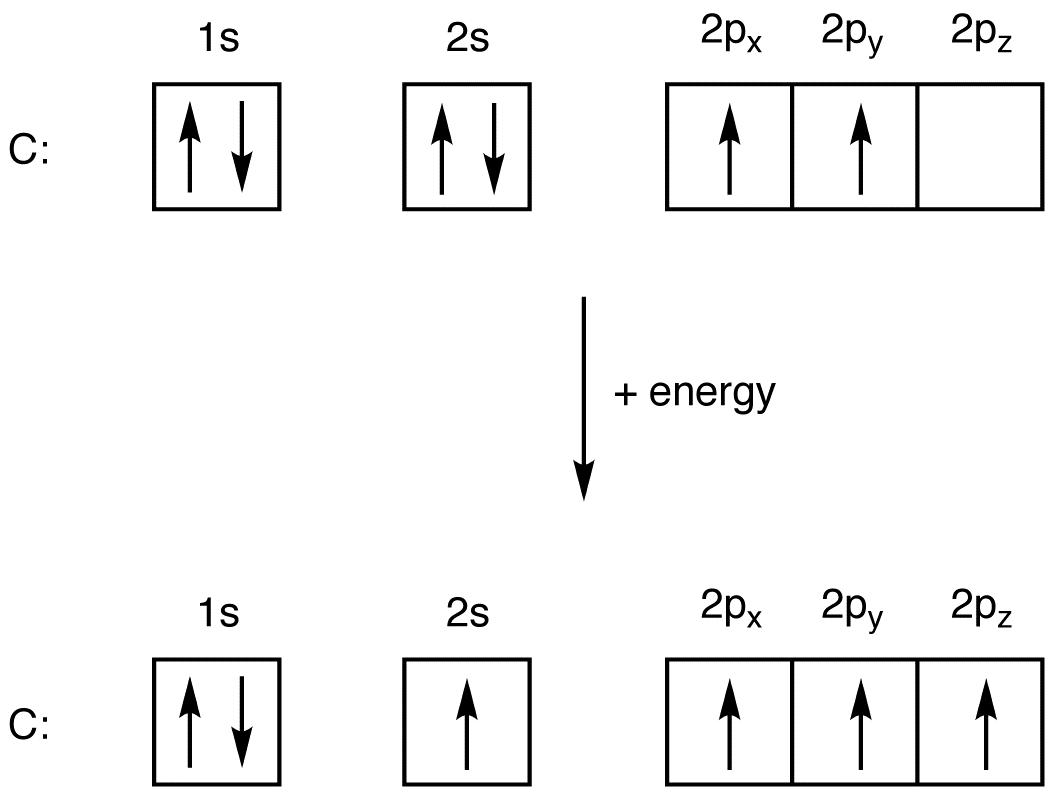
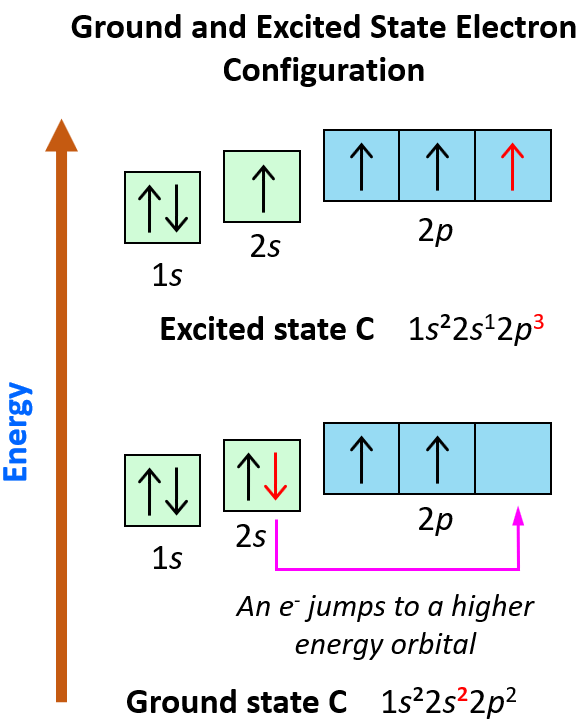
What is the excited state of carbon?
The Octet Rule The other halogen molecules (F 2, Br 2, I 2, and At 2) form bonds like those in the chlorine molecule: one single bond between atoms and three lone pairs of electrons per atom. This allows each halogen atom to have a noble gas electron configuration.
PPT Orbital Filling Electron Configurations PowerPoint Presentation
Hello Guys,Determining the electron configuration of any element is an easy and quick process, given that you know all the required information and general c.

Electronic Configuration for Carbon spdf Trick Chemistry Atomic
The electron configuration and the orbital diagram are: Following hydrogen is the noble gas helium, which has an atomic number of 2. The helium atom contains two protons and two electrons. The first electron has the same four quantum numbers as the hydrogen atom electron ( n = 1, l = 0, ml = 0, ms = +12 m s = + 1 2 ).
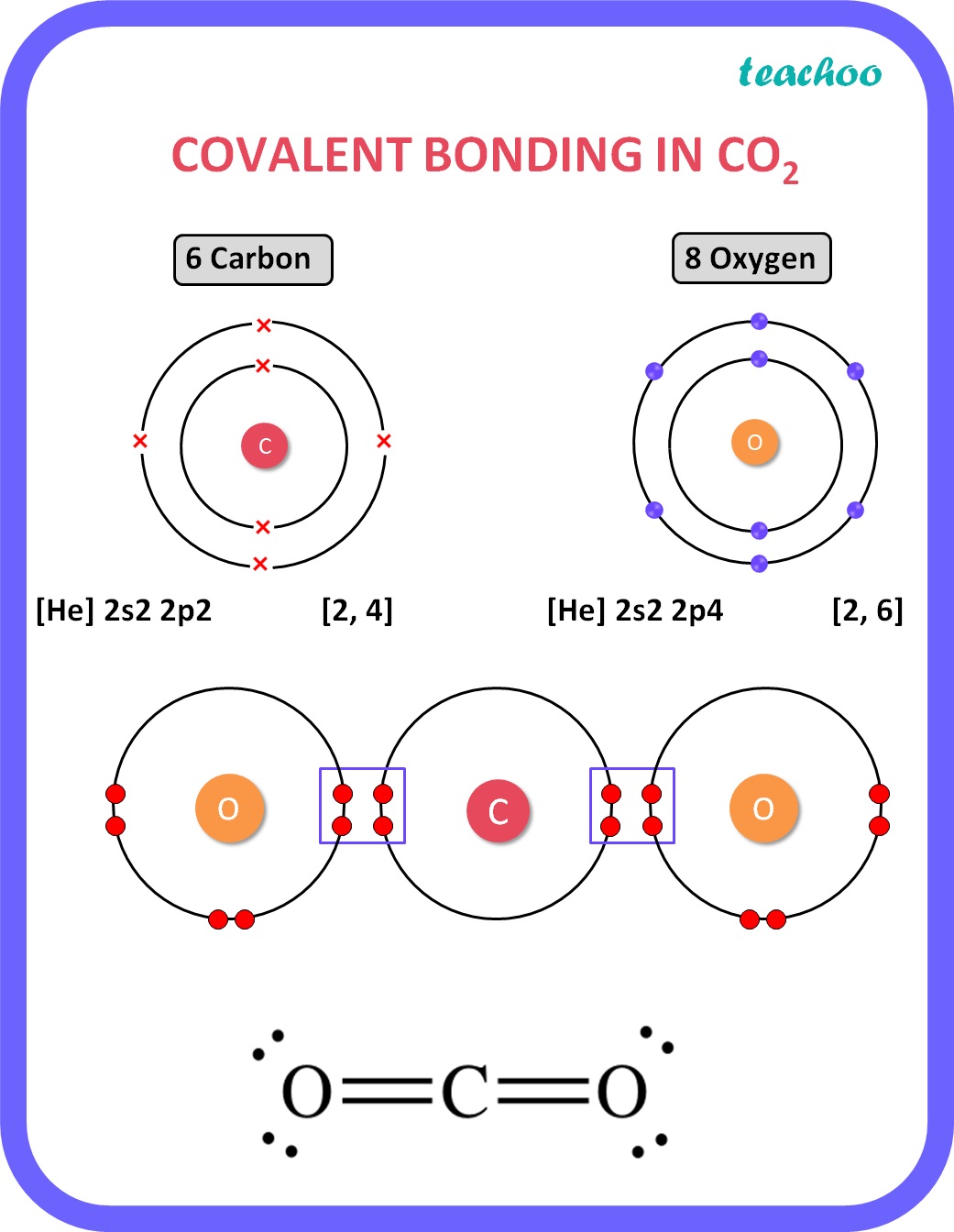
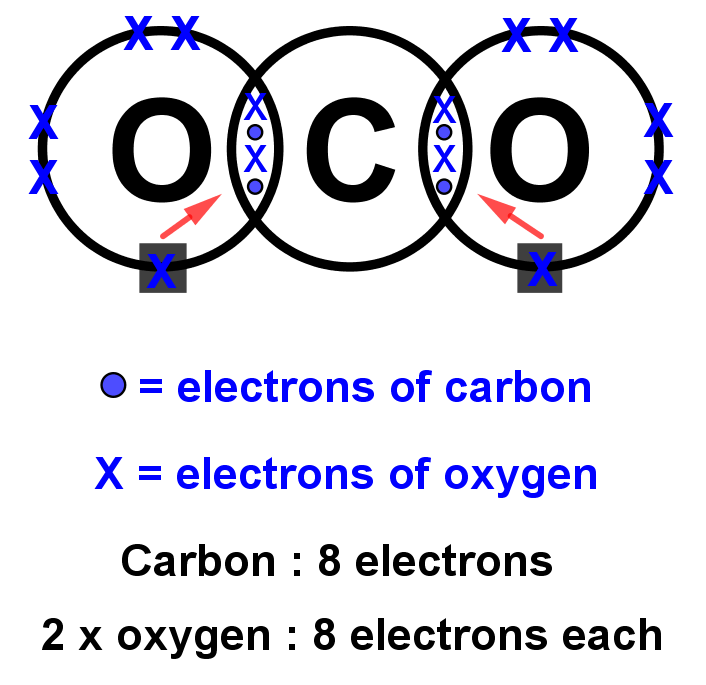
[Class 10] Electron dot structure of carbon dioxide with formula CO2
Answer: The electron configurations of the elements are presented in Figure 2.2.3, which lists the orbitals in the order in which they are filled. In several cases, the ground state electron configurations are different from those predicted by Figure 2.2.1. Some of these anomalies occur as the 3 d orbitals are filled.

Carbon Electron Shell Diagram
The n + l n + l rule tells you the order in which atomic orbitals are filled, and according to the rule the 4s 4 s orbital is occupied before the 3d 3 d orbital because it has lower energy. Thus, the electron configuration of Mn M n is [Ar]3d54s2 [ A r] 3 d 5 4 s 2 while that of Co C o is [Ar]3d74s2 [ A r] 3 d 7 4 s 2 .
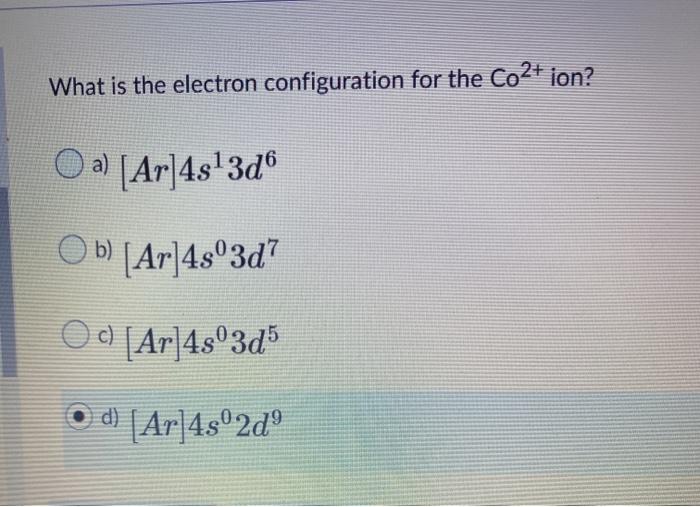
Solved What is the electron configuration for the Co2+ ion?
To determine the number of valence electrons for CO2, the Carbon dioxide molecule, we'll use the Periodic Table. Organizing the Periodic Table by Group, ski.
Carbon Electron Configuration Photograph by Photo
To write electron configuration of an element, locate its symbol in ADOMAH Periodic Table and cross out all elements that have higher atomic numbers. For example, if you need to write electron configuration of Erbium (68), cross out elements 69 through 120. Notice numbers 1 through 8 at the base of the table.
40 co3+ orbital diagram
The electron configuration of Cobalt is [Ar]4s 2 3d 7. When observing Cobalt 3+, we know that Cobalt must lose three electrons. The first two to go are from the 4s orbital and Cobalt becomes:[Ar]4s 0 3d 7. Then, the next electron leaves the 3d orbital and the configuration becomes: [Ar]4s 0 3d 6. Thus, we can see that there are six electrons.